Why Do Some People Have a Worse Response to the Coronavirus?

Wei-Shin Lai, M.D. June 8, 2020
Scientists are investigating why some people get sicker from the coronavirus than others. Every now and then we hear about a young, previously healthy person who dies from COVID-19. Those of us who fall under that category may wonder what else is a risk factor. There are the well-known risk factors, but here are some lesser-known reasons still under investigation.
Some ideas under investigation include the following:
1. Genetic differences impact on disease
- ABO blood type
- ACE2 receptor
- Biological sex
2. Antibody-Dependent Enhancement (ADE) caused by previous exposure to other coronaviruses
3. Exposure dose
4. Different strains of the novel coronavirus
Genetic Differences Impact on Disease
 ABO Blood Type
ABO Blood Type
A new very large and comprehensive study out of Italy and Spain from June 2020 looked at nearly 2,000 hospitalized patients and found that people with blood type A are most likely to be hospitalized. People with blood type O have a lesser chance of severe disease. This corroborates an initial study out of China with the same findings, but this study’s approach is from a completely different angle, which really solidifies the blood type association.
Details of the Italy and Spain ABO Blood Type Study
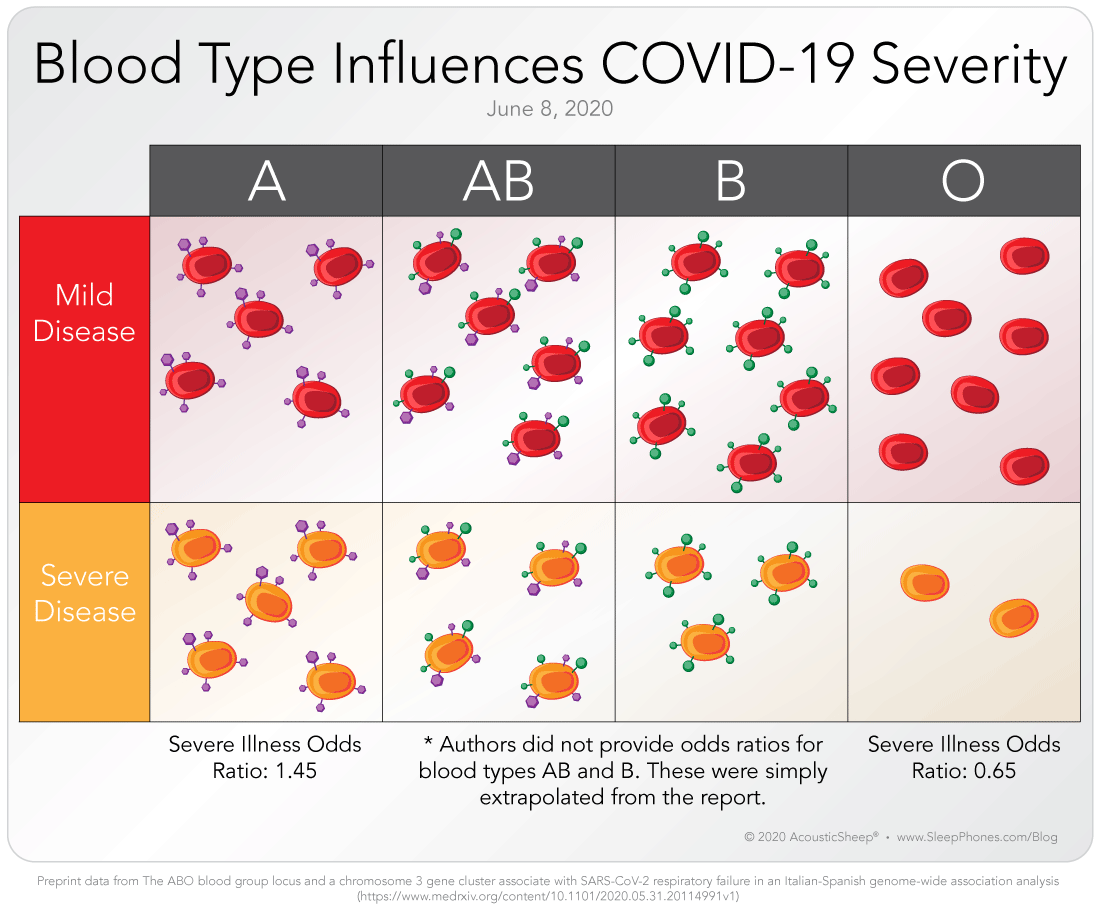
The Italy and Spain genomics testing is an incredible study. It is still pre-print and not peer-reviewed, but it has an incredibly long list of contributing authors and is a true international effort. The scientists were able to recruit nearly 2,000 patients and over 2,000 controls to contribute their blood for a genetic screening test. The scientists used a commercial genetic screening test that looked at over 700,000 locations in our genes, and through mathematical analysis identified only two locations that make a difference in severity of illness. The blood type location is on chromosome 9. It should be noted that the difference may not strictly be the actual blood cell antigen causing the problem but possibly the genes that are in the area of the area coding for the blood type antigen, which include associations with IL-6 (inflammatory marker), von Willebrand factor (related to coagulation), and Factor VIII (related to coagulation), according to the authors. The other is at the location of six different genes, and it is unclear yet which gene is implicated. Interestingly, it includes a gene associated with a transport protein that interacts with the ACE2 receptor, which is the exact receptor the virus binds to for introduction into cells.
ACE2 Receptor
There have been many studies characterizing how the ACE2 receptor impacts disease course. The ACE system is well-studied because it’s involved in high blood pressure, and there are medications that inhibit ACE (which is different from ACE2). What’s interesting is that despite all of the research on the ACE2 receptor, there has not been a clear “yes-or-no” on whether taking ACE inhibitors can affect outcome or whether ACE/ACE2 genetic variants cause worse disease. It seems that children have fewer ACE2 receptors in their nose compared with adults, which may explain the difference in susceptibility and disease outcomes, but it is simply an association, not proof.
Biological Sex
For a genetics study, I would have liked to see whether chromosome 23 (male versus female) impacts disease. It was not specifically called out in the Italian-Spanish study, and that’s perhaps because it did not meet statistical significance. There have been multiple studies indicating men having worse disease than women, so I’m curious if there was any trend supporting that.
Antibody-Dependent Enhancement (ADE) Caused by Previous Exposure to Other Coronaviruses
ADE stands for antibody-dependent enhancement. It’s a bad thing. Studies on SARS (the old one, not SARS-CoV-2) and MERS showed that ADE was a potential mechanism for worse illness. ADE has not been shown yet for COVID-19, although studies are underway. It’s important because it impacts vaccine safety. Basically, ADE is where if you have existing antibodies from other coronaviruses, those antibodies actually change how your body responds. Instead of making fresh new antibodies to neutralize the virus, your body recycles old antibodies that don’t neutralize the virus but still activates an overactive inflammatory response called a cytokine storm. Cytokines are supposed to help your body clear infections, such as raising your body temperature and summoning immune defenses. But in this case, they hit all at once and make a patient feel very sick very quickly. In the case of vaccine development, we need to avoid creating non-neutralizing vaccine-induced antibodies that actually result in ADE. That is where most of the research on ADE is focused right now.
Could Someone Who Had COVID-19 Once Get It Again and Develop Worse Disease From ADE?
It’s unlikely because if they were able to clear the disease the first time, then they likely produced effective antibodies. Those effective antibodies would last at least months to a few years. Previous research on SARS showed that people who had SARS continued to have detectable antibodies for 2-3 years afterwards. People who got better once would likely clear it even more quickly if exposed again within 2-3 years. Nothing’s ever a guarantee though. There are many reports of exceptions - people who have low antibodies, people who relapse later, people who have not yet cleared the infection after 3 months - but those are not the typical case.
Exposure Dose
There are reports of how healthcare providers seem to have worse illness than someone who catches the disease in the community. The theory is that they have a high exposure dose of the virus. Say they encounter 10,000 virus particles rather than 100. Those 10,000 would get a foothold and start replicating. The body’s immune system would attempt to respond. But someone who started with fewer viruses would take a bit longer to reach the same level of infection, while their body used that extra time to mount a response. The evidence is based on research on SARS (the old one) and influenza. Healthcare workers wearing more protective PPE (for example, N95 with a full-body gown compared with a surgical mask and a trash bag) faired better, because in theory, they had a lower exposure dose. There is no way to prove this directly because it’s unethical to deliberately infect people. Animal studies of various viruses have shown that exposure dose (infectious dose) seems to be important.
Different Strains
The different strains of the novel coronavirus may have different mortality rates, though there has been debate on this. The Nextstrain website has been collecting genomic information on the virus (looking at the exact RNA sequence) throughout the world. At this time, it’s basically just monitoring the viral mutations and scientists attempt to categorize them. If the virus picks up a mutation that causes it to be more or less fatal in one area of the world, then scientists can then attempt to figure out which mutations caused it. Previously, there was talk about a less deadly S-strain in China and a more deadly L-strain in Europe. There was also a pre-print paper saying that mutations have made the virus spread easier. Neither of these are widely accepted yet.
What We Do Know About Disease
While these are interesting ideas, and the ABO blood type differences seem to be very well substantiated, we cannot forget some of the clearest observations. Older people have worse disease. Pre-existing conditions like heart disease, high blood pressure, diabetes, obesity, and kidney disease all increase risk of worse disease. Unfortunately, we cannot change our blood type, lose weight too quickly, or turn back time. The things that we can actively do include reducing our chances for exposure, and if symptomatic, to talk to our doctor about starting a blood-thinning diet.
All product and other information obtained through or presented on the Site is for informational purposes only. You should consult with a qualified healthcare professional for specific information about any personal care, healthcare or medical needs. The products provided through and the information provided on the Site are not intended to substitute in any manner for medical treatment, medical advice, or medical diagnosis by a doctor or other healthcare professional. You should not forego, change or stop any tests or treatments that have been prescribed or recommended by any of your healthcare provider(s) without first consulting with your healthcare provider(s). You should not rely on any product you receive through or any information you receive from the Site when making any personal care, healthcare or medical decision, including but not limited to any decisions about treatment, testing, diagnosis or follow up. IF YOU ARE, OR SUSPECT THAT YOU ARE EXPERIENCING A HEALTH EMERGENCY, PLEASE CALL 911.
Brought to you by AcousticSheep LLC, maker of SleepPhones®, the most comfortable headphones you can wear in bed. SleepPhones® were invented by the author and her husband. Click here for more information. Dr. Lai also blogs about the coronavirus, having almost joined the CDC 15 years ago. 2020 © AcousticSheep LLC

